Dissociation constant (K_a) of formic acid and acetic acid are 2.5xx10^-4 and 0.5xx10^-5 respect... - YouTube

Electronically excited states of formic acid investigated by theoretical and experimental methods - ScienceDirect

What is the pH of a 0.15 M solution of formic acid, HCOOH ? `{:("Formic Acid ",K_a),(HCOOH - YouTube

Dehydrogenation of Formic Acid by a RuII Half Sandwich Catalyst - Vatsa - 2021 - ChemistrySelect - Wiley Online Library
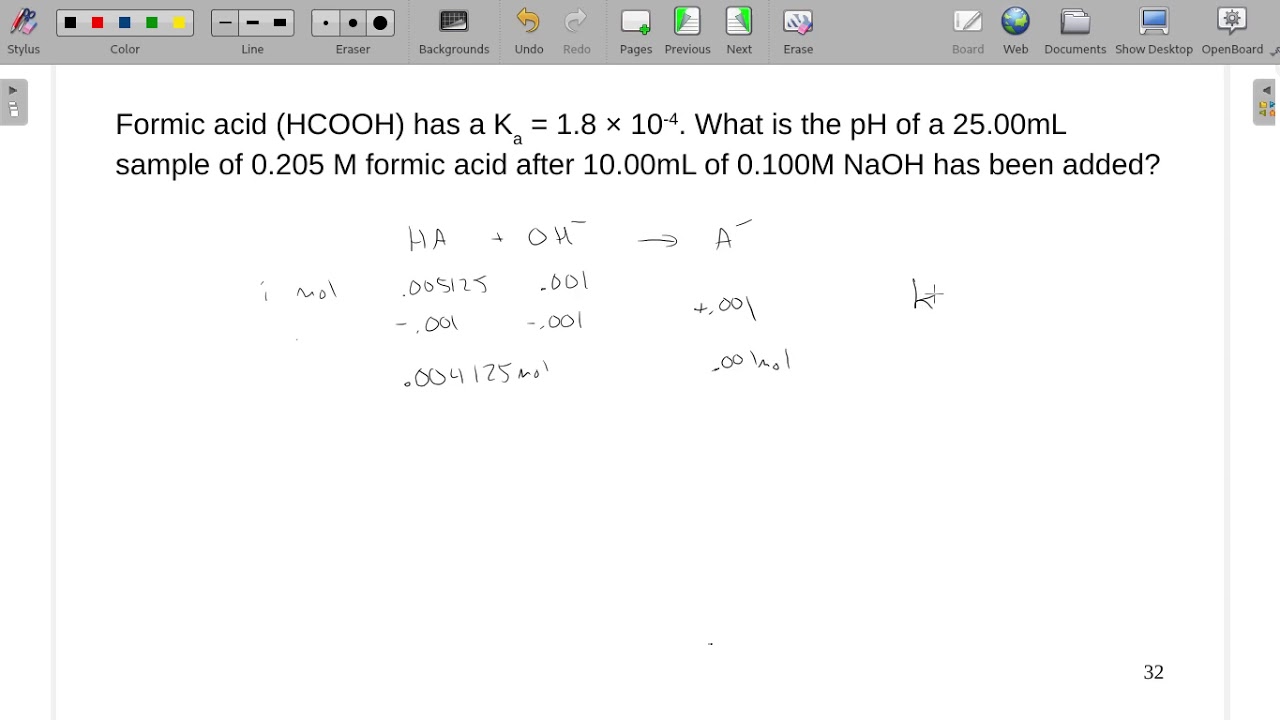
Formic acid has a Ka = 1.8×10-4. What is the pH of a 25.00mL sample of 0.205 M formic acid after... - YouTube

The Ka for formic acid is 18 x 10-4 What is the pH for a 035 M aqueous solution of sodium form - YouTube

Carbon Dioxide Hydrogenation to Formic Acid with Self‐Separating Product and Recyclable Catalyst Phase - Ehmann - 2022 - ChemCatChem - Wiley Online Library

Reaction and separation system for CO2 hydrogenation to formic acid catalyzed by iridium immobilized on solid phosphines under base-free condition - ScienceDirect
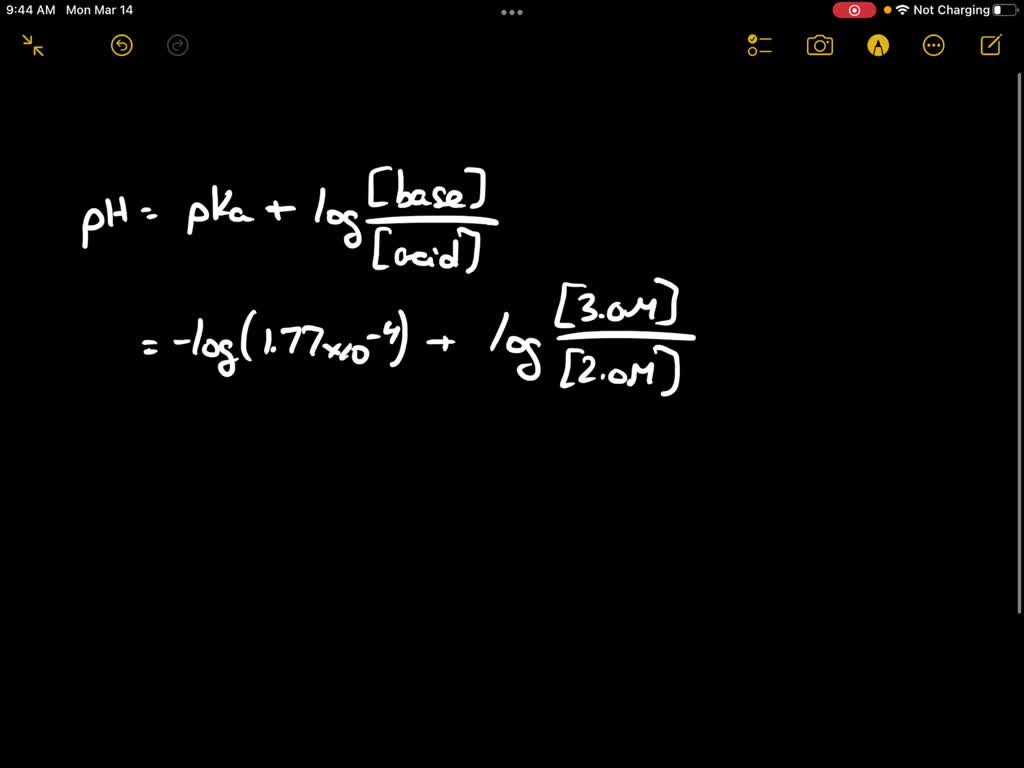
SOLVED: The Ka for formic acid, HCOOH, is 1.77 * 10^-4. HCOOH (aq) + H2O (l) ⇌ HCOO- (aq) + H3O+ (aq) What is the pH of a buffer made from 2.0

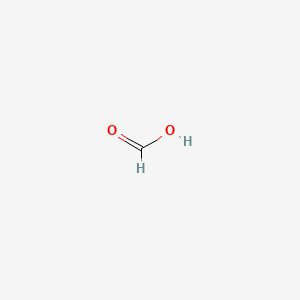
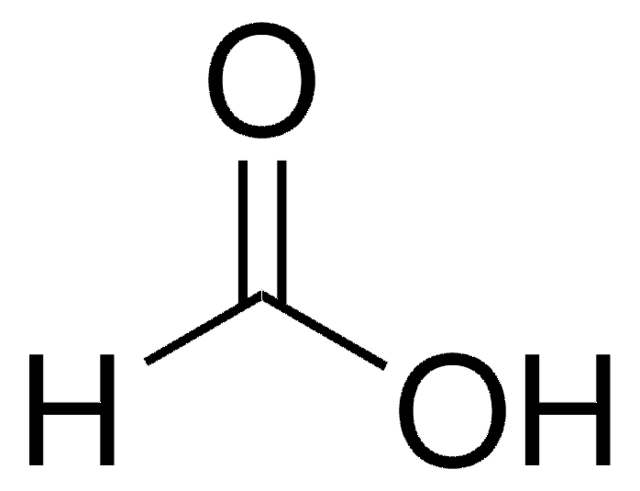
Formic Acid (HCOOH) - Structure, Molecular Mass, physical Properties, chemical properties, Uses and FAQs of formic acid (HCOOH)

Energies | Free Full-Text | Cost Efficiency Analysis of H2 Production from Formic Acid by Molecular Catalysts

Complex Mechanism of the Gas Phase Reaction between Formic Acid and Hydroxyl Radical. Proton Coupled Electron Transfer versus Radical Hydrogen Abstraction Mechanisms | Journal of the American Chemical Society






![a. A 0.1M solution of Formic acid [HCOOH] has Ka=1.77×10−4. Calculate (i).. a. A 0.1M solution of Formic acid [HCOOH] has Ka=1.77×10−4. Calculate (i)..](https://storage.googleapis.com/filo-classroom-notes/thumb_classroom_27777169_812PT.jpeg)